 Bulk Water with Exclusion Zone Water Characteristics: Experimental Evidence of Interaction With a Non-physical Agent
Bulk Water with Exclusion Zone Water Characteristics: Experimental Evidence of Interaction With a Non-physical Agent
Dibble WE1*, Kaszyk J2, Tiller WA1
1The William A. Tiller Institute, 909 S. Pinecone St., Payson, AZ 85541, USA
2Kasz Enterprises, Inc., PO Box 13835, Palm Desert, CA 92255, USA
*Correspondence E-mail: wdibble526@gmail.com
Key Words: EZ water, UV absorbance, Light scattering, Subtle energy
Received Oct 28th, 2013; Revised Dec 13th, 2013; Accepted Dec 15th, 2013; Published March 20th, 2014; Available online March 25th, 2014
Abstract
The UV absorption spectrum of exclusion zone (EZ) water has been used to define the unique signature of this type of water structure. Specifically, an absorption peak at a wavelength of 270 nm is found to be characteristic of EZ water. We present experimental evidence that this same behavior can be found in certain bulk waters. This includes waters not contaminated by organic substances that are known to absorb UV radiation at this wavelength.
We present UV absorption data for bulk water treated using a remote treatment method (both before and after treatment) that shows the characteristic 270 nm peak only after such treatment. In addition, an inflection point (shoulder) at 210 nm is found which is a new spectroscopic feature for this type of treated water. These treated waters also show unique and characteristic light scattering features that have not been reported before. Anomalous turbidity enhanced by the input of mechanical energy appears to be characteristic of these treated waters.
Article Outline
- Introduction
- Materials and Methods
- Results
- Discussion
- Conclusions
- References
- Discussion with Reviewers
Introduction
This paper is not about exclusion zone (EZ) water specifically, but about a kind of bulk water that has some characteristics resembling EZ water. Explicitly, the UV absorption peak at 270 nm, characteristic of EZ water [Chai et al. (2008)], is shown to be present in a bulk water that has been treated in a specific way (remotely). Experiments we have performed show that water structure heretofore limited to the zones near solid surfaces may be present in bulk water as well. Besides the anomalous UV absorption, other optical properties of treated bulk water are also observed including anomalous light scattering. These two newly described characteristics of remotely treated bulk water are almost certainly related.
Relevant Exclusion Zone Water Behavior
Depending on the specific wall material isoelectric point of a container holding a specific aqueous solution, it is well known that various electrolyte species kinetically redistribute in the near-wall region to ultimately produce thermodynamic electrochemical potential equilibrium throughout the container. The thickness of this wall screening, Debye-length, can be quantitatively determined from the aqueous solution electrolyte composition and specific wall adsorbates via solution of the Poisson Equation.
Very small, well-characterized, spherical polystyrene latex beads have been used experimentally as sensor probes [Zheng and Pollack (2003), Zheng et al. (2006) and Chai et al. (2008)] of this near-surface region. These latex beads relocate in this general experimental system because of their different isoelectric point surface potential relative to that of the container wall. Net repulsion of the latex beads from the container wall has been experimentally observed and defines a wall-region exclusion zone (EZ). The water in the EZ has also been experimentally observed to produce a UV absorption peak at a wavelength of 270 nm. The magnitude of this peak decreases as one approaches the bulk fluid where no UV absorption peak at this wavelength is generally found.
Bulk Water with EZ Water Characteristics
There are natural bulk waters that show enhanced UV absorption sometimes with peaks near 270 nm. Armstrong and Boalch (1961) [Figure 2] found that both Irish Sea and Firth of Clyde seawater show UV absorption peaks near 270 nm. Normal seawater and artificial seawater do not show the enhanced UV absorption or the 270 nm peaks. The filtered Clyde water was visibly yellow and the enhanced UV absorption was attributed to organic contamination (Armstrong and Boalch, 1961). Organic contamination producing 270 nm UV absorption peaks was also a concern of Chai et al. (2008). Thus, care must be taken to consider contamination when evaluating natural bulk waters that may show a 270 nm UV absorption peak.
If organic contamination can be ruled out, some other agent must be responsible for a 270 nm peak possibly present in the UV absorption spectrum of any given natural bulk water. Ideally, the water could be tested before the agent was applied and then after the agent was used. If the 270 nm peak only occurs after the agent was applied, then the agent could reasonably be assumed to be the cause. We use a proprietary agent remotely to produce the 270 nm UV absorption peak in bulk naturally occurring water (Arrowhead Spring Water). [In principle, this allows anyone with a UV spectrometer to send us Arrowhead Spring Water in sealed containers, have us apply the remote treatment agent and then have us return the sealed water containers for UV absorption studies to be done by others.]
Besides UV absorption spectroscopy, we use quantitative nephelometry to observe the light scattering qualities of these treated bulk waters. Nephelometry involves the study of the scattering of light by a medium containing light-scattering particles. In water, light-scattering particles cause a cloudy appearance technically called turbidity. Using a device that measures water turbidity, we have found that waters treated remotely with (a proprietary) subtle energy input, can scatter light when untreated controls do not. The water that exhibits the UV absorption peak at 270 nm also shows anomalous light scattering. We have also found that a mechanical energy input can have a strong influence on this light-scattering effect as well as UV light absorbance.
Materials and Methods
Treated Arrowhead Spring water is spring water obtained from a commercial source and treated remotely using a proprietary method. We shall, for convenience, label this commercial effect as a “subtle energy” effect. Kaszyk created a process using proprietary catalytic towers that work energetically to interact with matter when it is coupled with the subtle energy field. The result is that Kaszyk unknowingly concentrated a new form of subtle energy that can be infused into liquids remotely. To test these treated waters in our lab, we sent to Kaszyk’s lab water samples of our own, in sealed containers, to eliminate any possible contamination. The returned samples could then be evaluated with some confidence that no conventional physical or chemical changes had occurred.
UV Absorption Spectroscopy
The measurement instrument used for all studies is the Cole-Parmer UV/Visible Spectrophotometer, 115 VAC, 60 Hz (Part Number EW-83059-10). The resolution is 1 nm. Quartz glass cuvettes were used and Grade 1 purified water was used as a blank. The procedure followed was to set the wavelength and then measure and set the blank followed immediately by a measurement on the sample. A series of measurements from 200 nm to 320 nm took less than one hour to complete for 1 nm wavelength intervals. For each wavelength, the absorbance and % transmission values were recorded manually and then entered into an Excel spreadsheet. Commercial Arrowhead Spring Water was used in both the treated and untreated studies.
Nephelometry
When light is passed through a suspension of solid particles, scattering dissipates part of the light and part is transmitted directly through the medium. Measurement of the intensity of light transmitted is related to the concentration of the phase responsible for the scattering. The units of measurement are called nephelometric turbidity units (NTUs). Nephelometric analysis is most sensitive for very dilute suspensions. In fact, the government-established water quality standards require turbidity values for drinking water to be below 1 NTU and in many cases below 0.3 NTU. As a consequence of these water quality standards, modern nephelometers need to be highly sensitive and accurate at these low turbidity levels.
The measurement device we use is a Hach 2100Q nephelometer that uses a tungsten lamp light source (Web Reference). The lower detection limit of the device is about 0.02 NTU for water at resolution of .01 NTU. The uncertainty in the readings is ±0.01 NTU. The uncertainty was determined by measuring our purified water standard many hundreds of times over a year. This purified water (Grade 1, ISO 3696, 1987) was obtained from our in-house water purifier at different times over this year period. The turbidity values were always in the range 0.03±0.01 NTU using this purified water. For general water sample measurements, we averaged over 5 readings. The device itself averages over 3 readings (default mode) so, for each measurement, an average of over 15 readings was performed for all the experiments for each reported data point.
Calibrations of the nephelometer were performed regularly using calibration standards of constant NTU (turbidity) value. Measurements of control solutions were also performed regularly. These measurements also showed little to no changes over a period of several months. There was no problem obtaining and maintaining usable control solutions for this purpose that have very low NTU values over long periods of time (ranging from 0.02 to 0.04 NTUs for our purified water and 0.04 to 0.05 NTUs for the Arrowhead water).
Results
Results – UV Absorption Spectroscopy
The UV absorption of bulk treated and fresh Arrowhead water has been examined over a wavelength range of 200 to 320 nm. This is all in the UV range of the electromagnetic spectrum with the 320 nm end of the range approaching visible light. The results are presented in Figure 1.
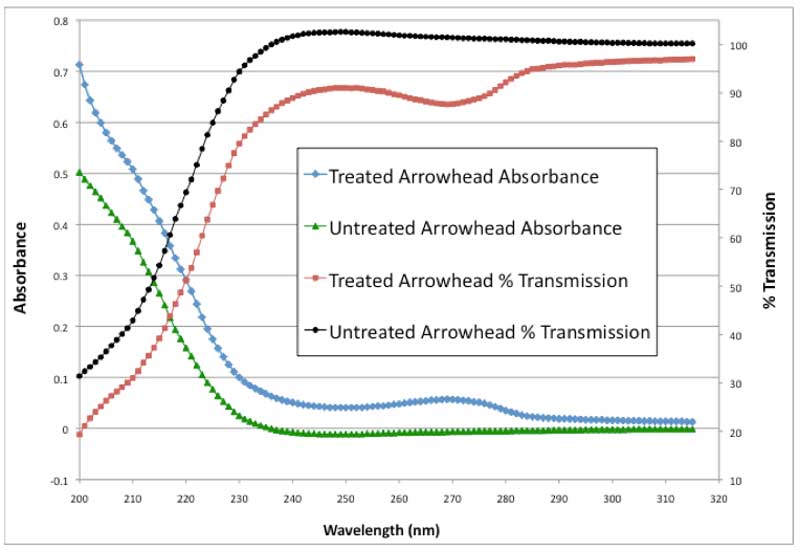
Figure 1: UV absorbance and % transmission vs. wavelength.
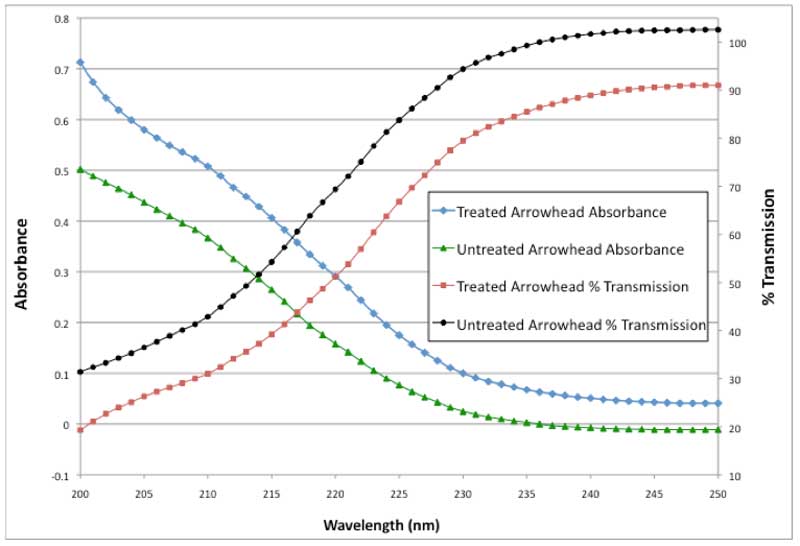
Figure 2: UV absorbance and % transmission vs. wavelength (detail: 200-250 nm).
As can be seen in Figure 1, there is a substantial absorption peak at 270 nm similar to EZ water as described by others (Chai et al., 2008). In addition, there is a pronounced shoulder or inflection point at 210 nm. This inflection point has not been reported before for either bulk water or EZ water. A close up of this shoulder is shown in Figure 2. In the untreated water absorbance, the curve from 200-215 nm is concave downward. In the treated water absorbance up to the shoulder, the curve is concave upward indicating the presence of a shoulder near 210 nm. In Figure 1, the treated absorbance values are all higher than the untreated values. The two curves tend to approach each other as the visible wavelengths are approached. Thus, anomalous UV absorption is demonstrated for treated water at all UV wavelengths.
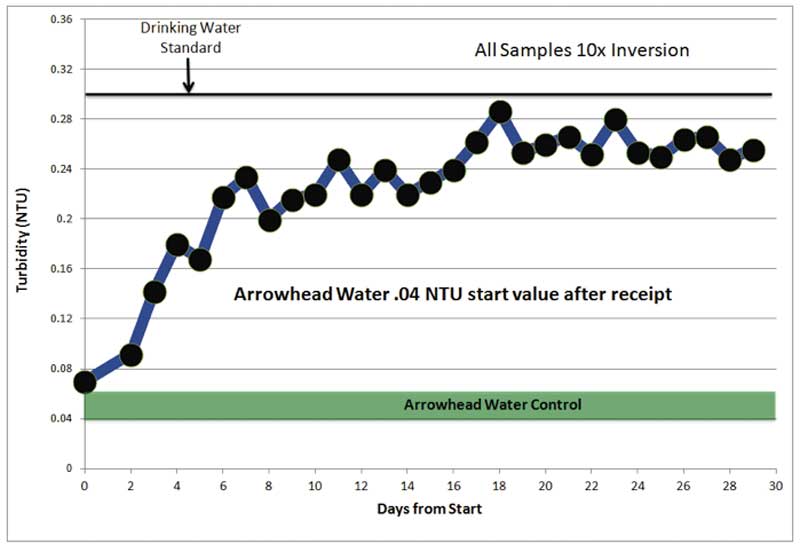
Figure 3: Turbidity (NTU) vs. time.
Results – Nephelometry
Using a nephelometer to measure light scattering from treated vs. untreated Arrowhead water is also very revealing. The Arrowhead Spring water was remotely exposed to Kaszyk’s proprietary catalytic towers. This procedure produced treated water using an effect we call a “subtle energy” effect. We measured the turbidity values of our own purified water as well as locally acquired Arrowhead Mountain Spring water from a commercial vendor. These water samples were placed in glass bottles that would eventually be sent to Kaszyk for treatment. After we were satisfied with the baseline turbidity readings that we were obtaining from these controls, we sealed the tops of the bottles with liquid wax. After the wax hardened, we ink-marked the surface of the wax and sent the bottles to Kaszyk. When the treated bottles were returned, we carefully verified that the wax seal was undisturbed. In two test cases, we sent and received the bottles via mail. In one case, we hand-delivered the bottles to Kaszyk and returned to our lab with them after Kaszyk’s treatment. After unsealing the returned, treated bottles, we made turbidity measurements immediately. The first two tests were inconclusive. The reason for this was revealed by the third test. When the 3rd sealed bottle was returned, we inverted the sample before each measurement in a change of protocol that had not been used in the earlier sealed bottle tests. In this case, like the other sealed bottles, the control Arrowhead water sample had not been tampered with during the treatment process and the initial value measured (before inversion) was close to the value that it read before sending to Kaszyk. However, thereafter, the sample was inverted prior to measurement and the result shown in Figure 3 was obtained.
The Figure 3 result was a substantial new discovery that indicated that the input of mechanical energy enhanced the light scattering effect. Note that the effect is cumulative with time and that the values approached those that are considered high for drinking water. At this point we realized that the Kaszyk treatment process does significantly affect our sealed control waters; but to measure it we needed to activate the sample via a mechanical inversion process. This inversion process resembled a re-suspension of light-scattering particles even though there were, apparently, no light-scattering solid particles physically added to the sealed sample.
We were concerned that air bubbles (nanobubbles) may have been contributing to the increase in turbidity values in the sealed sample cells after many inversions; and we, therefore, performed vacuum experiments on these samples. These tests showed that this was not a concern as before and after vacuum results were not much different. We were also concerned that physical particles may have been contributing to the positive results from the many tests on commercial samples that we had performed and we conducted studies on these using a centrifuge. Centrifugation tests carried out on the commercial samples indicated that no solid particles had been added.
Kaszyk’s commercial product, Sun Lover’s Mist (SLM), is Arrowhead Spring Water after Kaszyk’s proprietary treatment has been performed. SLM is delivered from a container via the use of a spray nozzle. The nozzle breaks the fluid into small droplets that appear as a mist. For our experiments, discussed above, the fluid was taken directly from the glass or plastic bottle containers. Considering the profound effects relatively small mechanical energy inputs had on the measurement results, we wondered what affect the spray delivery method would have. We designed an experiment involving filling a nephelometer sample-cell with Sun Lover’s Mist using the spray nozzle delivery method and measuring the turbidity directly afterwards.
Our first concern was that too many gas bubbles would make the results ambiguous. However, the degassing procedure, mentioned above, could be used to remove gas bubbles as well as dissolved gas. No gas bubbles were seen forming when Sun Lover’s Mist was sprayed onto the side of the sample cell. The sprayed fluid tightly coated the wall and drained down the side of the cell into the bulk fluid. The fluid had a visual appearance that was unusual. It’s viscosity when it runs down the walls appeared greater than normal water, almost oily. After the cell was filled with Sun Lover’s Mist in this fashion, measurements began immediately.
Results of nephelometric measurements showed values higher than we had seen before, reaching as high as 3 NTUs or more (nearly 100 times control values). After vacuum application and some time, the values steadied at just above 1 NTU or higher than the highest level allowed for drinking water. Over a period of days these values decayed to 0.5 NTU or still higher than any value seen previously and an order of magnitude greater than control values. The values could be restored to nearly 1 NTU by merely gently inverting the sample cell 3 times.
These nephelometry results made us wonder if the mechanical energy input might affect UV absorption as well. This was necessary to verify, as it is very important for evaluating the quality and stability of the commercial product. Therefore, we also performed an experiment where we sprayed Kaszyk’s SLM product into a UV spectrometer cuvette. The results are shown in Figure 4 that compares the unsprayed product with the sprayed product. The absorbance values for the sprayed sample are higher over the whole wavelength range measured showing that the mechanical energy input did have an influence on UV absorbance.
Discussion
The issues raised by the results presented above fall into two categories. The first issue is what are the UV spectroscopic and light scattering instruments actually measuring. The second issue is, once we understand the first issue, what are the measurements telling us about this particular water that resembles EZ water in several important ways.
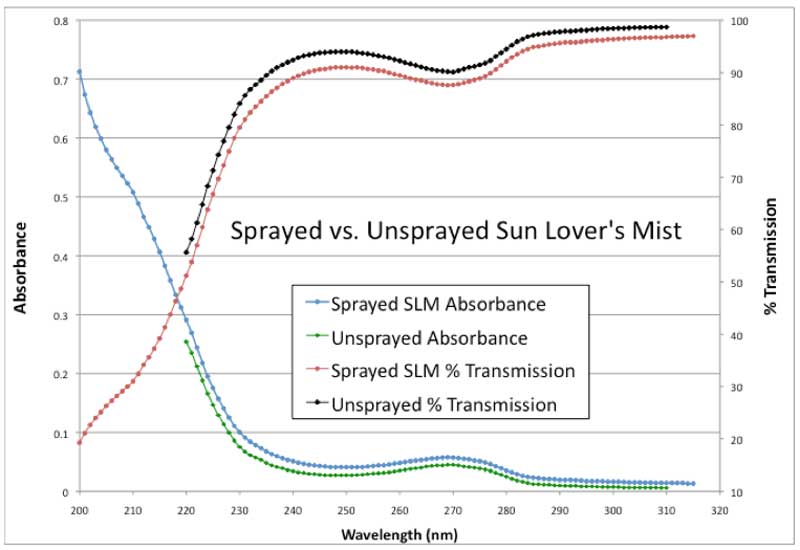
Figure 4: UV absorbance and % transmission vs. wavelength.
Measurement Issues – Nephelometer
The way our particular nephelometer wo-rks, it is not possible to distinguish light emitted by a sample from the light scattered by solid particles in the sample. If particle-sized domains of the sample emitted light, it would appear as light scattering to our nephelometer. Light emission from such domains would be measured as higher turbidity values. If this happened with conventional water samples, nephelometry would be useless for determining particle density or turbidity. Since nephelometry can be performed on conventional waters with low NTU values (low turbidity) with extremely high reliability and linearity with respect to particle standards, we can conclude that such light emission does not normally occur. The subtle energy-treated samples with high NTU values exhibit an unambiguous Tyndall effect (using a laser beam). If light emission is involved, it mimics scattering extraordinarily well.
Measurement Issues – UV Spectrophotometer
The way spectrophotometers work, light at a particular wavelength is partially absorbed (or partially transmitted) through a sample. This energy absorption is caused by the interaction of the light with the material medium. For absorbed light, energy from the light is absorbed in the volume of the material. Particulate matter residing in the medium causes light scattering and this scattering changes the direction of the light. So, for scattered light, the light emerges in a different direction from the incident light. If light is substantially scattered by particulate material in the sample, a spectrophotometer measuring absorption (via the Beer-Lambert Law) will erroneously assign the scattering that reduces the light intensity to absorbance. Turbidity in a sample will, thus, lead to errors in interpretation of the absorbance results produced by a spectrophotometer.
Then What Are We Actually Measuring?
Spectrometers measure the light absorbed by a sample at a given wavelength. If the sample scatters light, the spectrometric measures are underestimated, which leads to errors in their interpretation. However, the absorbance peak at exactly 270 nm in Figures 1 and 4 is the precise absorbance peak for EZ water. This correspondence alone would suggest that something very real is being measured. If scattering were somehow producing these results, why would that particular absorbance peak be singled out?
For our measurements using a UV spectrophotometer, the wavelengths of light measured are in the UV range. For the turbidity measurements using a nephelometer, the wavelengths of light used are in the visible range; so, on the surface, it appears that the two measurement results may not relate to one another. However, it is not uncommon for real UV absorption in a sample to lead to visible wavelength photon emission in the same sample (fluorescence). Possible fluorescence represents an interesting potential explanation for anomalously high turbidity measurements using our nephelometer.
In summary, the treated samples are anomalous in that the bulk treated water appears to absorb UV photons peaking at 270 nm. Untreated water does not do this. The treated water is also unusual in that the water exhibits highly anomalous turbidity (light scattering effects). Untreated water does not exhibit this anomaly either. Significantly anomalous behavior using two different measurement methods has been demonstrated using this non-locally treated water.
Conclusions
The input of mechanical energy appears to enhance both UV absorption (Figure 4) and light scattering (Figure 3) for treated samples. This is a significant discovery. Further experimental work that needs serious consideration includes light scattering studies in the UV wavelength range and light emission studies in the visible range. If non-locally treated waters exhibit enhanced scattering of light in the visible range as revealed by a simple Tyndall effect test, then maybe our turbidity measurements reveal something real and are not due to anomalous light emission only. The possibility exists that these structured waters both absorb in the UV and scatter in the visible but do not absorb and scatter together over any particular wavelength range.
Some kind of extraordinary water structure has been revealed in bulk water treated remotely using subtle energy transmission without the use of any form of EM radiation. This structure appears to be nearly identical to that of EZ water that is structured by the proximity to solid surfaces (Zheng and Pollack, 2003 and Zheng et al., 2006). From our work, based on the time we have been evaluating these waters, this structure appears to be stable for very long periods of time (years) in the bulk. Paradoxically, the structuring appears to be enhanced by the input of mechanical energy in various forms. Mechanical energy input enhances these anomalies possibly in a similar way that light energy increases the exclusion zone thicknesses (Chai et al., 2009).
References
Armstrong, FAJ and Boalch, GT (1961) The Ultra-violet Absorption of Sea Water. J. Mar. Biol. Ass. U.K. 41: 591-597.
Chai BH, Zheng JM, Zhao Q, and Pollack GH (2008) Spectroscopic Studies of Solutes in Aqueous Solution. J. Phys. Chem. A 112: 2242-2247.
Chai B, Yoo H, Pollack GH (2009) Effect of Radiant Energy on Near-Surface Water. J. Phys. Chem. B 113: 13953-12958.
Zheng JM and Pollack GH (2003) Long-range forces extending from polymer-gel surfaces. Phys. Rev. E 68: 031408.
Zheng JM, Chin WC, Khijinak E, Khijinak Jr E, Pollack GH (2006) Surfaces and interfacial water: Evidence that hydrophilic surfaces have long-range impact. Adv Colloid Interface Sci 127: 19-27.
Web References
Discussion with Reviewers
Reviewer Smith CW: From your spread-sheet data you should able to do an audit covering the incident, scattered and transmitted light. The sum of the red and blue curves in Figure 1 may confirm or eliminate the possibility of absorption or fluorescence.
Dibble WE, Kaszyk J and Tiller WA: The scattered light is measured in the visible spectrum range. The absorbance characteristics are measured in the UV range so the measurements do not overlap. However, the suggestion of auditing the data is interesting. We took the raw % transmission data and multiplied by 100 giving the absolute transmission. We then calculated log10 of the absolute transmission to get absolute absorbance. We compared these values to the absorbance values the spectrophotometer produced and found a very close agreement as expected. We then added the absolute transmission and the absolute absorbance and found they do not sum to 1 exactly. Contrasting these sums for the treated water vs. the untreated water (subtracting one from the other) revealed further peaks and troughs in the resulting plot that we are still evaluating.
Reviewer: In the nephelometry experim-ents, the scattered light distribution should enable the size of the scattering identities to be determined. The scattered light should be polarised.
Dibble, Kaszyk and Tiller: This is a good point. The unit that we use does not allow for a ratio of the side scatter and the front scatter, which would be an indication of the size of the particle. We did however send samples to Hach Laboratories, the manufacturer of the nephelometer, for ratio testing and the results were interesting. First, their results exactly matched the scattering values we obtained. Also, they did a spectroscopic scan of the visible spectrum to rule out “contamination” and they showed no absorbance peaks across the entire visible spectrum showing that no “particles” were present. The ratio results showed significant forward scatter compared to side scatter and that would support some kind of “large” structure. We did not do a polarized light study, but that is planned for a future experiment to show birefringence, which would indicate a crystalline type structure.
Reviewer: The spectrometer has put a frequency signature from the cell into the 270 nm peak from where information can be extracted. It matches a ‘sunscreen product’.
Dibble, Kaszyk and Tiller: The marker at 270 nm is associated with ring type structures. This falls into the part of the UV spectrum known as UVC which is not typical for most sunscreen products.
Reviewer: Your ‘spray’ experiment implies that making a droplet smaller than a coherence domain does not erase its information which reappears on collection in the cell.
Dibble, Kaszyk and Tiller: The spray experiment is at first counterintuitive. It has led us to consider additional experiments to show that Subtle Energy in the sample causes the formation of a new kind of droplet, perhaps very similar to that described by Dr. Pollack in his book, “The Fourth Phase of Water”. If that is true, then the droplet acts like a tiny battery with a separation of charge caused by the negatively charged shell and the protons captured within the droplet as it forms. It may be that adding mechanical energy helps to form a large cluster that changes the structure and observable visible flow of the liquid due to the organizing potential (information content) of Subtle Energies.
Reviewer: Your Conclusion seems to imply that the input of mechanical energy (i.e. succussion, a momentum impulse which potentises) can enhance UV absorption and light scattering. It presumes that subtle energy is not related to EM radiation. Reference to one of your U(1)/SU(2) papers might be appropriate.
Dibble, Kaszyk and Tiller: Subtle energy produces many anomalies that characterize its presence. Our working hypothesis is that we observe the enhanced UV absorption and light scattering because this organizing energy (through addition of information) could explain these observations.
